MiRXES Pte Ltd |
Accelerate Your Biomarker Discovery |
Biomarker discovery studies are paving the way to a new paradigm of a predictive, preventive and patient-centric model of clinical care. Finding accessible, accurate, specific and sensitive biomarkers will enable early detection of diseases, guide treatments, predict disease progression and ultimately save lives. Discovering, validating and profiling biomarkers can be time and resource intensive. To streamline and accelerate the development and implementation of reliable and robust biomarkers, standardized methodologies spanning sample collection, transport, storage, experimental protocols and data normalization and analyses need to be implemented. MiRXES provides comprehensive miRNA profiling services in our dedicated high-throughput biomarker discovery laboratory. Our tested and proven workflow has generated high impact papers and one of the world’s earliest miRNA IVD assays for early gastric cancer detection. Extensive pre-analytics and quality control measures ensure data quality and reliability. We adhere to standardized methodologies published in our national standard for the design, development and validation of miRNA-based diagnostics1. 1. SS 656:2020 Singapore Standard, Design, Development and Validation of miRNA-based diagnostics. Enterprise Singapore. |
 |  |
 |  |
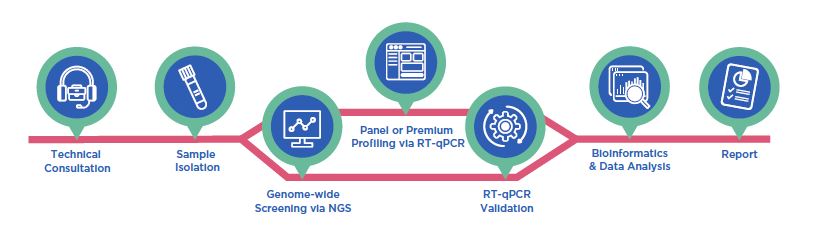
Comprehensive miRNA Biomarker Discovery and Validation Services Tailored to Your Project Goals |
| ID3EAL Premium Profiling Service |
|
| ID3EAL Panel Profiling Service |
|
| Genome-wide Screening Via NGS |
|
 |
Request for a Free Trial To speak to our profiling expert, contact us at sales@mirxes.com |




